BAMBUTEROL HCL
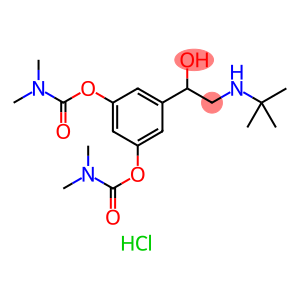
Bambuterol Hydrochloride
CAS: 81732-46-9
Molecular Formula: C18H30ClN3O5
BAMBUTEROL HCL - Names and Identifiers
| Name | Bambuterol Hydrochloride |
| Synonyms | BAMBEC KWD-2183 Bambuterol HCl BAMBUTEROL HCL BAMBUTEROL HYDROCHLORIDE Bambuterol Hydrochloride Bambuterol monohydrochloride 5-[2-(tert-butylamino)-1-hydroxyethyl]benzene-1,3-diyl bis(dimethylcarbamate) dimethylcarbamic acid 5-[2[(1,1-dimethyl)amino]1-hydroxyethyl]-1,3-phenylene ester hydrochloride DIMETHYLCARBAMIC ACID 5-[2-[(1,1-DIMETHYLETHYL)AMINO]-1-HYDROXYETHYL]-1,3-PHENYLENE ESTER HYDROCHLORIDE Carbamic acid, dimethyl-, 5-(2-((1,1-dimethylethyl)amino)-1-hydroxyethyl)-1,3-phenylene ester, monohydrochloride |
| CAS | 81732-46-9 |
| EINECS | 643-060-9 |
| InChI | InChI=1/C18H29N3O5/c1-18(2,3)19-11-15(22)12-8-13(25-16(23)20(4)5)10-14(9-12)26-17(24)21(6)7/h8-10,15,19,22H,11H2,1-7H3 |
| InChIKey | LBARATORRVNNQM-UHFFFAOYSA-N |
BAMBUTEROL HCL - Physico-chemical Properties
| Molecular Formula | C18H30ClN3O5 |
| Molar Mass | 403.9 |
| Density | 1.154g/cm3 |
| Melting Point | 222-224°C |
| Boling Point | 500.9°C at 760 mmHg |
| Flash Point | 256.7°C |
| Solubility | H2O: ~33mg/mL, soluble |
| Vapor Presure | 7.49E-11mmHg at 25°C |
| Appearance | powder |
| Color | off-white |
| Merck | 14,953 |
| Storage Condition | Inert atmosphere,Room Temperature |
| Refractive Index | 1.533 |
BAMBUTEROL HCL - Risk and Safety
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| HS Code | 29242990 |
BAMBUTEROL HCL - Standard
Authoritative Data Verified Data
This product is 1-[bis-(3 ',5'-N,N-dimethylcarbamoyloxy) phenyl]-2-n-tert-butylaminoethanol hydrochloride. Calculated as dry product, containing no less than 98.5% of C18H29N305 • hc1.
BAMBUTEROL HCL - Trait
Authoritative Data Verified Data
- This product is white or off-white crystalline powder; Odorless.
- This product is soluble in water or methanol, soluble in ethanol or chloroform, almost insoluble in ethyl acetate or acetone.
BAMBUTEROL HCL - Differential diagnosis
Authoritative Data Verified Data
- take about 20mg of this product, add 5ml of water to dissolve, and add 5 drops of ammonium chromate thiocyanate to generate light red precipitate.
- take an appropriate amount of this product, add water to dissolve and dilute to make a solution containing about 0.40mg per 1 ml, and measure it by UV-Vis spectrophotometry (General rule 0401), there is a maximum absorption at a wavelength of 263nm and a minimum absorption at a wavelength of 238nm.
- The infrared absorption spectrum of this product should be consistent with that of the reference product (General rule 0402). If not, take the appropriate amount of this product and the reference product, add acetone-water (6:1) solution to dissolve, put it in an ice bath, wait for crystallization and filter it, after filtration and drying under reduced pressure at 50 ° C., the sample was measured again.
- the aqueous solution of this product was chloride identification (1) of the reaction (General 0301).
BAMBUTEROL HCL - Exam
Authoritative Data Verified Data
acidity
take 0.20g of this product, Add 10ml of water to dissolve, and measure according to law (General rule 0631). The pH value should be 4.5~7.0.
clarity and color of solution
take 0.20g of this product and add 10ml of water to dissolve, the solution should be clear and colorless; If it is turbid, it should not be deeper compared with No. 1 turbidity standard solution (General rule 0902 first method); If it is colored, comparison with yellow No. 2 Standard Colorimetric solution (General rule 0901 first method), not deeper.
Related substances
take this product, add mobile phase to dissolve and dilute to make a solution containing about 1 mg per 1ml as a test solution; Take 1ml for precision measurement and put it in a 100ml measuring flask, dilute to the scale with the mobile phase, shake, and serve as a control solution. Test according to high performance liquid chromatography (General 0512). Silica gel was bonded with eighteen alkyl silane as filler; [methanol-acetonitrile-phosphate buffer solution (take potassium dihydrogen phosphate 6.8g, add appropriate amount of water to dissolve, adjust pH to 3.0 with phosphoric acid, dilute to 1000ml with water)(30:18:52), containing 1.5g of sodium octane sulfonate per 3000 ml] as mobile phase; The detection wavelength was 214nm; The number of theoretical plates was not less than based on the bambuterol peak, the degree of separation of bambuterol peaks from adjacent peaks shall be as required. 20 u1 of the test solution and the control solution were respectively injected into the human liquid chromatograph, and the chromatogram was recorded to 2 times of the retention time of the main component peak. If there are impurity peaks in the chromatogram of the test solution, the area of a single impurity peak shall not be greater than 0.5 times (0.5%) the area of the main peak of the control solution, and the sum of the areas of each impurity peak shall not be greater than the area of the main peak of the control solution (1.0%).
loss on drying
take this product, dry to constant weight at 105°C, weight loss shall not exceed 0.5% (General rule 0831).
ignition residue
take l.Og of this product and check it according to law (General rule 0841). The residue left shall not exceed 0.1%.
Heavy metals
take this product l. Add 2ml of acetate buffer solution (pH 3.5) and appropriate amount of water to 25ml, and check according to law (General rule 0821, first method), containing no more than 15 parts per million of heavy metals.
BAMBUTEROL HCL - Content determination
Authoritative Data Verified Data
take this product 0.3g, precision weighing, add glacial acetic acid 20ml, Mercury acetate test solution 5ml to dissolve, add crystal violet indicator solution 1 drop, with high gas acid titration solution (0.1 mol/L) titration to the solution of pure blue, and the titration results with blank test correction. Each 1 ml of perchloric acid titration solution (0.1 mol/L) corresponds to 40.39mg of C18H29N305. HCl.
BAMBUTEROL HCL - Category
Authoritative Data Verified Data
B-receptor agonists.
BAMBUTEROL HCL - Storage
Authoritative Data Verified Data
light shielding, sealed storage.
BAMBUTEROL HCL - Bambuterol Hydrochloride Tablets
Authoritative Data Verified Data
This product contains bambuterol hydrochloride (C18H29N305 • HCl) should be 90.0% ~ 110.0% of the label amount.
trait
This product is white or off-white.
identification
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- take an appropriate amount of fine powder of this product, add water to dissolve and dilute to make a solution containing about 0.4mg of bambuterol hydrochloride per 1 ml, filter, and take the filtrate, as determined by UV-Vis spectrophotometry (General rule 0401), there is a maximum absorption at a wavelength of 263mn and a minimum absorption at a wavelength of 238nm.
- Take appropriate amount of fine powder of this product, add water, shake, filter, filtrate visible gasification product identification (2) reaction (General rule 0301).
examination
- relevant substances: take an appropriate amount of fine powder of this product (equivalent to 50mg of bambuterol hydrochloride), put it in a 50ml measuring flask, add an appropriate amount of mobile phase, and shake to dissolve bambuterol hydrochloride, dilute to the scale with mobile phase, shake, filter, and take the filtrate as the test solution; Take 1ml with precision, put it in a 100ml measuring flask, dilute to the scale with mobile phase, and shake well, as a control solution. According to the chromatographic conditions under the content determination item, accurately measure 20ul of each of the above two solutions, respectively inject human liquid chromatography, record the chromatogram to 3 times the retention time of the main component peak. If there are impurity peaks in the chromatogram of the test solution, the sum of each impurity peak area shall not be greater than 1.5 times (1.5%) of the main peak area of the control solution. The content uniformity of
- shall be calculated from the content of each tablet measured under the content determination item, and shall comply with the regulations (General rule 0941).
- the dissolution of this product, according to the dissolution and release determination method (General rule 0931 second method), water 900ml as the dissolution medium, the speed of 50 rpm, according to the law, after 15 minutes, 10ml of the solution was taken, filtered, and the filtrate was taken and diluted with water to prepare a solution containing 10ug of bambuterol hydrochloride per 1 ml as a test solution; in addition, take an appropriate amount of bambuterol hydrochloride reference substance, weigh it precisely, dissolve it with water and dilute it quantitatively to make a solution containing about 10ug per lml, which is used as a reference solution and determined according to the method under the content determination item, the dissolution amount of each tablet was calculated. The limit is 80% of the labeled amount and shall be in accordance with the provisions.
- others shall be in accordance with the relevant provisions under the item of tablets (General rule 0101).
Content determination
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using eighteen alkyl silane bonded silica gel as filler; [methanol-acetonitrile-phosphate buffer (take potassium dihydrogen phosphate 6.804g, add water 800ml to dissolve, adjust the pH value to 3.0 with phosphoric acid, add water to make 1000ml, shake well)(30:18:52), containing 1.5g of sodium octane sulfonate per ml as mobile phase; The detection wavelength is 214nm. The number of theoretical plates shall not be less than 3000 based on the bambuterol peak.
- determination Method: Take 10 tablets of this product, put them in 50ml measuring flask respectively, add appropriate amount of water, fully shake to dissolve bambuterol hydrochloride, dilute to the scale with water, shake and filter, take appropriate amount of continuous filtrate with precision and dilute it quantitatively with water to make a solution containing about 40ug of bambuterol hydrochloride in each lml, which is used as a test solution. Take 20u1 injection of human liquid chromatograph with precision quantity, record the chromatogram; Take an appropriate amount of bambuterol hydrochloride reference substance, weigh it precisely, dissolve it with water and dilute it quantitatively to make a solution containing about 40ug per lml, and determine it by the same method. The content of each tablet was calculated by peak area according to external standard method, and the average content of 10 tablets was obtained.
category
with bambuterol hydrochloride.
specification
(l)10mg (2)20mg
storage
light shielding, sealed storage.
BAMBUTEROL HCL - Reference Information
| Overview | Bambuterol hydrochloride Hy droochloride has the chemical name (R, S)-1-[BIS -3 ', 5'-(N, N-dimethylcarbamoyloxy) phenyl]-2-n-terbutoxyethanol hydrochloride for the treatment of asthma, one of the main drugs for emphysema and bronchitis, and has the effect of protecting the fetus and lowering blood lipids in the first trimester. It has a long duration of action as a bronchial antispasmodic anti-asthmatic drug, and side effects are very weak, without significant effects on the heart and digestive system. |
| Use | for the treatment of bronchial asthma, asthmatic bronchitis, obstructive emphysema and other pulmonary diseases accompanied by bronchospasm. Ester prodreg of the 2-adrenaline agonist Terbutaline (T109750). Bronchodilator. |
| synthesis method | with 3 ', 5'-dihydroxyacetophenone (2) as the starting material, after esterification, bromination, condensation, pressure hydrogenation reduction, salt preparation 1. Preparation of 3 ', 5'-bis (N, N-dimethylcarbamoyloxy) acetophenone (3) 3 ', 5'-dihydroxyacetophenone (2) (15.2g,0.1mol), anhydrous potassium carbonate (27G, 0.2mol) and ethyl acetate (100ml), N, N-dimethylcarbamoyl chloride (32g ,0.3mol) were added at room temperature, after adding the mixture to reflux for 3 h, it was poured into water (150ml), and the organic layer was separated. The aqueous layer was extracted with ethyl acetate (50ml × 3), the organic layers were combined, washed with water until neutral, dried over anhydrous sodium sulfate, distilled off ethyl acetate, and recrystallized from petroleum ether (60-90 ° C.) to give 3. Preparation of 3 ', 5'-bis (N ,N-dimethylcarbamoyloxy)-ω-bromoacetophenone (4) 3(29.4g,0.1mol), dichloroethane (0.1 ml), slowly and Dropwise with stirring bromine (16g, mol) in dichloroethane (50ml) solution, add the reaction 2H at 25 °c, poured into water (ML) the organic layer was separated. The organic layer was washed with water until neutral, dried over anhydrous sodium sulfate, the solvent was recovered by distillation, and the residue was recrystallized from methanol (50ml) to give 4. Bambuterol hydrochloride (1) was added sequentially with 4(18.6g,0.055mol) and DMF(40ml), and sodium borohydride (0.93g,0.025mol) was added at room temperature, after reaction at 50 ° C. For 2H, the mixture was poured into water (100ml) and extracted with dichloromethane (50ml × 3). The organic layer was washed with water (50ml × 3), dried over anhydrous sodium sulfate, and the solvent was recovered to dryness. Add anhydrous ethanol (40ml), tert-butylamine (7.3g,0.1mol), Reflux reaction for 3H, add water (10% ml), adjust pH 10~12 with NaOH (about 30ml), extract with dichloromethane (50ml x 3). The organic layer was washed with deionized water (50ml x 3) and dried over anhydrous sodium sulfate. Dry hydrogen chloride gas was added to the solution at pH 2-4 and concentrated to dryness. Isopropanol (100ml) was added, heated to reflux, decolorized with activated carbon (about 1.0g) for 15min, filtered while hot, cooled to precipitate crystals, suction filtered and dried to obtain product 1. (2016-01-13) |
| attention should be paid to | 1. Contraindications ① allergic to this drug, Terbutaline and sympathomimetic amine. Idiopathic hypertrophic Subvalvular aortic stenosis. ③ patients with tachyarrhythmia. (4) liver cirrhosis or liver dysfunction should not be used. 2. Use with caution ① renal insufficiency. ②Patients with recent myocardial infarction. ③ patients with heart disease such as hypertension. ④ patients with diabetes mellitus. (5) patients with hyperthyroidism. Patients with increased sensitivity to sympathomimetic amines (such as uncontrolled hyperthyroidism). 3. Effect of the drug on children the dose for children under 12 years of age has not been established, infants and young children should be used with caution. 4. The effect of drugs on the elderly with liver, kidney and heart dysfunction in elderly patients with caution. 5. Effect of drug on pregnancy this drug relaxes uterine smooth muscle, can inhibit uterine activity and delivery, animal experiments have not found that the drug has teratogenic effect, pregnant women should be used with caution (especially in early pregnancy). Although the pregnancy safety of this drug has not been established, the pregnancy safety of Terbutaline, the active metabolite of this drug, is classified as B by the U. S. Food and Drug Administration (FDA). Effects of the drug on lactation Terbutaline is secreted into the milk but does not adversely affect the infant at the therapeutic dose. It has been reported that premature infants receiving beta 2 ~ adrenergic receptor agonist therapy in lactating women will produce temporary hypoglycemia, should be used with caution. |
| biological activity | Bambuterol HCl is a potent β-adrenopptor antagonist for the treatment of asthma. |
| Target | Value |
Supplier List
CAS: 81732-46-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 81732-46-9
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 81732-46-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 81732-46-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 81732-46-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 81732-46-9
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 81732-46-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 81732-46-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025



